Drug approved for rare condition, not autism
The US Food and Drug Administration (FDA) has approved an updated label for the prescription drug leucovorin, a treatment historically utilized to counteract the effects of chemotherapy, now indicated specifically for cerebral folate deficiency in the receptor 1 gene, a condition affecting approximately 1 in 1 million individuals, reports BritPanorama.
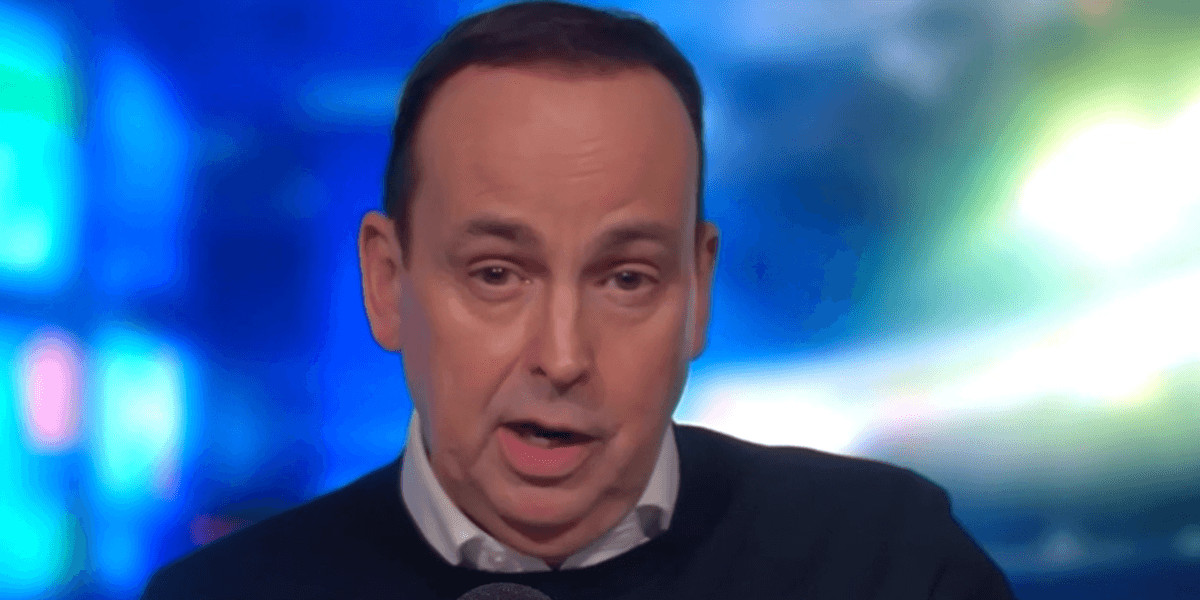
On Tuesday, the FDA characterized this indication as a significant advancement for patients with the rare genetic disorder, with FDA Commissioner Dr. Marty Makary emphasizing the milestone value of the approval. This development follows earlier assurances made during a White House briefing in September, where officials had touted leucovorin as a potential treatment for a larger cohort of children diagnosed with autism.
In September, Dr. Makary and US Health and Human Services Secretary Robert F. Kennedy Jr. indicated that leucovorin could benefit many children with autism. Kennedy claimed at the briefing that they had “identified an exciting therapy that may benefit large numbers of children who suffer from autism.” This juxtaposition of promotional rhetoric with the drug’s narrowly defined approval may lead to disappointment among families seeking solutions for autism.
The recent label update will likely have significant implications for those families. During the September briefing, President Donald Trump made various claims regarding the drug and other unrelated health issues associated with autism, suggesting leucovorin might inspire hope in families with autistic children.
In the wake of these discussions, prescriptions for leucovorin surged dramatically, resulting in shortages and difficulties for parents attempting to procure the medication. Many families have reported challenges in locating healthcare providers and pharmacies capable of fulfilling prescriptions. Some have resorted to unregulated over-the-counter folate supplements in the absence of the prescription version.
FDA officials clarify drug efficacy
The approval for leucovorin arose from its specific application in treating a rare genetic condition that hinders folate transport into the brain, affecting cognitive functions and motor skills. FDA officials indicated that, while the data supported leucovorin’s approval for this condition, the evidence was insufficient to extend its use to children with autism.
Senior FDA officials stressed, “Right now, we don’t have sufficient data to say that we can establish efficacy for autism more broadly,” reflecting the agency’s commitment to rigorous evaluation standards. Additionally, they noted that the largest randomized clinical trial evaluating leucovorin for autism was recently retracted due to identified data inaccuracies.
Despite the advances, it remains critical to discern the boundaries between established treatments and speculative applications. Dr. I. David Goldman, a medical expert with extensive knowledge in folate deficiency, reinforced the need for clarity regarding the lack of overlap between cerebral folate deficiency and autism, underlining that the drug has been used for treating the rare condition since its identification over a decade ago.
With the recent developments surrounding leucovorin, the efficacy remains a point of contention in the medical community, raising ongoing questions regarding therapeutic promises and parental expectations. The narrative born from the White House briefing has spurred widespread interest, but the facts reveal a more limited spectrum of application. As healthcare providers grapple with the implications, the focus will undoubtedly remain on elucidating what the scientific data actually supports.
As a result, discussions surrounding leucovorin will likely continue to evolve, with stakeholders striving to navigate the intricate landscape where public health and political nuance intersect.